GMJ Medicine
eISSN : 2626-3041
Volume 4, Issue 1 (2025)
GMJM 2025, 4(1): 1-5 |
Back to browse issues page
History
Received: 2024/06/3 | Accepted: 2024/11/22 | Published: 2025/01/18
Received: 2024/06/3 | Accepted: 2024/11/22 | Published: 2025/01/18
How to cite this article
Mohaghegh Daghigh G, Soeibipour L, Abakelia S. Antidepressant-Like Effects of Menthol via Dopaminergic System; Using Forced Swimming Test in a Mice Model. GMJM 2025; 4 (1) :1-5
URL: http://gmedicine.de/article-2-242-en.html
URL: http://gmedicine.de/article-2-242-en.html
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



Rights and permissions
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
1- Georgian Center for Neuroscience Research, International Center for Intelligent Research, Tbilisi, Georgia
Keywords:
| Abstract (HTML) (2686 Views)
Full-Text: (691 Views)
Introduction
Major depression is a complex psychiatric disorder and almost 20% of the people are faced with it during their lifetime [1]. It is related with signs such as mood, interest, feeling, hope, appetite, sleep, performance and social relationships [1]. It was reported that typical antidepressants encourage synaptic action in the monoamines including dopamine (DA) through blocking the neuronal reuptake of these monoamines [2]. Serra et al. [3] suggested that the involvement of dopaminergic presynaptic receptors in the antidepressants’ activity a long back, it was not mentioned as one of the accepted hypotheses of mental depression.
The impaired antioxidant system is associated with Reactive Oxygen Species (ROS) [4]. An increase in the production of ROS in depression is associated with increased levels of malondialdehyde and arachidonic acid [5]. It was reported an increase in oxidative stress in the animals submitted to stress [5]. It is accepted to use antioxidant agents for treatment of the depression [6].
Menthol is a strong antioxidant and improves the antioxidant and lipid profile in animals [7]. Abbasi-Maleki et al. [8] have reported that crude extract of Mentha piperita showed antidepressant-like effect through the monoaminergic system due to its compound such as menthol. As far as we know, no scientific study is conducted to evaluate the antidepressant-like effects of menthol by forced swimming test (FST) and through dopaminergic system using a mice model. The FST is broadly used in pharmacological models for evaluating the antidepressant effects [8].
The study was thus performed for evaluating the antidepressant-like effects of menthol via dopaminergic system in the FST model.
Materials and Methods
Animals
Male Swiss albino mice with weight of 22±2g and 6 weeks of age were used in this study. The animals were maintained in cages (n=4) with a lighting cycle 12h light/12h dark cycle, ambient temperature of 23-25°C and humidity of 55±15%. The feed and water were given ad libitum. The commercial pellet food was bought from Javaneh Khorasan, Iran.
Materials
Menthol, SCH 23390 and sulpiride were purchased from Sigma-Aldrich, St. Louis, MO, USA. Only SCH 23390 was subcutaneously (SC) administrated, but other agents and menthol were intra-peritoneally (IP) administered in a constant volume of 10ml/kg. The FST was conducted 1h after the single injection of the chemicals and menthol.
Acute toxicity test
We used the Lorke's method for investigating toxicity test [9]. Briefly, after 24h of fasting, a total number of twenty NMRI mice were divided into 5 groups (n=4) and intra-peritoneally treated with vehicle (normal saline with 10% Tween-80; 10ml/kg) and menthol (10, 15, 20, and 30mg/kg). The animals were monitored for behavioral and toxicity signs and also mortality for 24h.
Forced Swimming Test
The FST as a pharmacological test is used for evaluating the antidepressant activity of the agents. We conducted the FST as reported by Porsolt et al. [10]. 28 NMRI mice were grouped into seven groups as follows:
- Group I = vehicle (with 10% Tween 80; 10ml/kg, IP);
- Groups II-V = menthol (10, 15, 20, and 30mg/kg; IP, respectively); and
- Groups VI-VII = fluoxetine (20mg/kg, IP) and imipramine (30mg/kg, IP)
The immobility, swimming and climbing were evaluated as stated by previous studies [8] for the rest four minutes during a six-minute period. We considered decreased immobility time and increased climbing or swimming times as behavioral profiles consistent with antidepressant-like activity [10].
Dopaminergic system of menthol antidepressant-like
To evaluate the potential involvement of the dopaminergic system in the antidepressant-like activity of menthol, the mice, were pre-treated with SCH 23390 (0.05mg/kg, SC, a dopamine D1 receptor antagonist), sulpiride (50mg/kg, IP, a dopamine D2 receptor antagonist) and haloperidol (0.2mg/kg, IP, non-selective dopamine receptor antagonist) 15min before the IP administration of menthol (20mg/kg, IP) or vehicle (10ml/kg, IP). The animals were exposed to FST 1h after the treatment [8].
Statistical analysis
The data were analyzed by the one-way ANOVA for dose-response curves and time-course curves and two-way for assessing of the mechanism of action followed by Tukey's post-hoc test when appropriate.
Findings
Acute toxicity test
There were no any mortality and toxicity signs in the mice studied with doses used (10-30mg/kg, IP).
Immobility behavior
Our findings for one-way ANOVA in Figure1 showed a significant reduction in immobility time (p<0.001) and a significant increase in swimming time (p<0.001) in the mice treated with menthol compared with the vehicle group. There were significant differences in the immobility time caused by menthol compared to that produced by treatment of imipramine and fluoxetine. The one-way ANOVA did not also show significant differences between the mice treated with menthol (20 and 30mg/kg, IP) and fluoxetine in swimming time (p>0.05) and also between the animals administrated with menthol (30mg/kg, IP) and imipramine in climbing time (p>0.05). The best response was observed in dose of the 20 and 30mg/kg dose, this dose was thus used 20mg/kg dose for the subsequent tests (Figure 1).
Involvement of the dopaminergic system in the antidepressant-like effect of mentholin the FST
Pre-treatment of the animals with SCH 23390 (a dopamine D1 receptor antagonist), sulpiride (a dopamine D2 receptor antagonist) and haloperidol (non-selective dopamine receptor antagonist) significantly prevented the antidepressant-like effect of menthol (Figure 2).
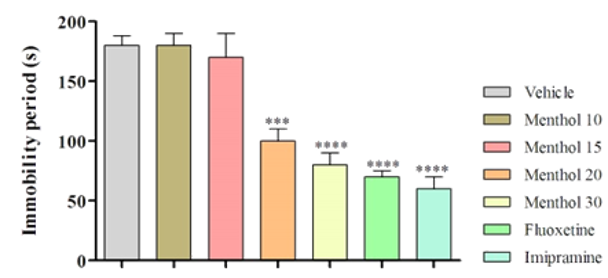
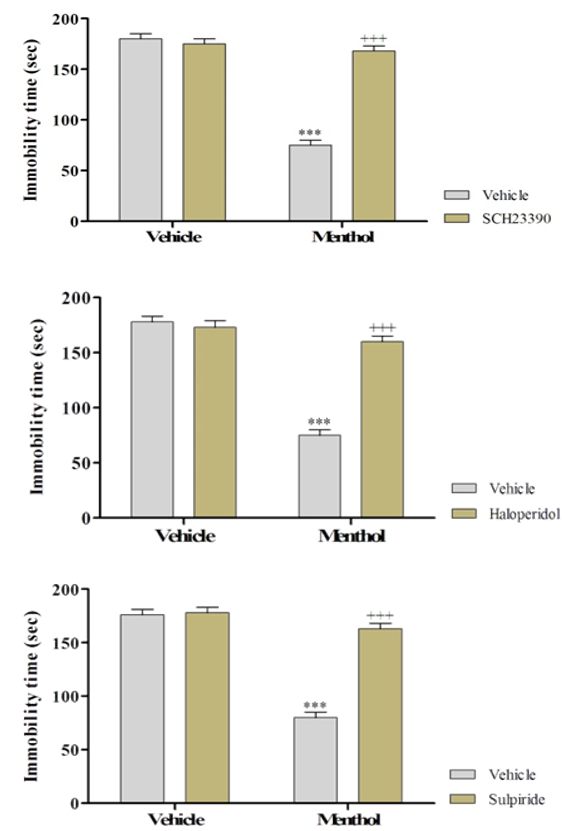
Figure 2. The effect of the pretreatment of animals with SCH23390, sulpiride and haloperidol on the antidepressant-like effect created by the menthol in the FST (***significant differences among other groups and the control at p<0.001; +++significant differences among other groups and the extract group at p<0.001)
The OFT
It was not observed significant changes (p>0.05) in the crossing (Figure 3) and rearing counts in the animals treated with menthol (10-30mg/kg; IP) in comparison to the vehicle group.

Figure 3. The effect of the menthol in OFT in the mice. Values are expressed as mean ± S.D.
Discussion
This study was conducted to evaluate the antidepressant-like effects of menthol by FST through dopaminergic system in mice model. In the current study, the results showed that menthol administered through i.p. route yields the antidepressant-like effect in the FST in mice. In addition, the locomotor activity was not altered. In addition, an involvement of dopaminergic (D1 and D2 receptors) systems in the antidepressant-like effect of menthol was observed. The FST is commonly used as a behavioral test and also a model for evaluation of the depression in rodents [10]. The results showed that the effect of the menthol at dose of 30mg/kg in the FST was similar to the response of fluoxetine and imipramine. It was reported that typical antidepressants encourage synaptic action in the monoamines including dopamine (DA) through blocking the neuronal reuptake of these monoamines [2]. It has been accepted the essential role of monoaminergic system in the patho- physiology and treatment of depression [2].
Traditional antidepressants decrease the depression signs by increasing the bioavailability of monoamines in the brain [11, 12]. The antidepressants have been used for the treatment of depression, because of increased synaptic levels of these monoamines [13]. It seems that menthol increases the levels of monoamines as is evidenced by increased dopaminergic levels. It has been reported that the antidepressant-like effects of active compounds such as menthone [14], cineole [15] and pulegone [16]. There is no any report available in the literature for antidepressant-like effects of menthol. Abbasi-Maleki et al. [8] attributed antidepressant-like effects of medicinal plants to their antioxidant properties. The results showed that menthol acts in the antidepressant-like effects through dopaminergic system. Antidepressant-like effects of the menthol were reversed through the pretreatment of animals with SCH23390, sulpiride, and haloperidol suggesting that menthol exerts antidepressant-like effects via the dopaminergic system. It has been accepted the role of the D1 and D2 receptors in the treatment of depression, because antagonists of these receptors reverse the antidepressant-like action of some antidepressants [17-19]. The dopamine directly promotes the factors diminished in people with depression such as motivation and attention [20]. It was reported that some dopamine receptor agonists have positive roles in the treatment of refractory and bipolar depression [21]. The results showed that dopamine D1 and D2 receptor antagonist reversed the antidepressant-like effect of the menthol showing that dopamine D1 and D2 receptors mediate the antidepressant-like effect of menthol. The results also showed that pretreatment of mice with haloperidol could reverse the antidepressant-like effect of the menthol, suggesting that dopamine receptors modulate in antidepressant-like response of the menthol.
Conclusion
The administration of menthol exerts antidepressant-like effects through dopaminergic (D1 and D2) system.
Acknowledgements: None declared by the authors.
Ethical Permission: Compliance with ethical guidelines: Approval for this study was obtained from International Center for Intelligent Research.
Conflicts of Interests: None declared by the authors.
Authors' contributions: All authors contributed toward data analysis, drafting and revising the paper and agreed to be responsible for all the aspects of this work.
Funding/Support: This study was supported by a grant from Georgian Center for Neuroscience Research (GCNR-2018-1841335).
Major depression is a complex psychiatric disorder and almost 20% of the people are faced with it during their lifetime [1]. It is related with signs such as mood, interest, feeling, hope, appetite, sleep, performance and social relationships [1]. It was reported that typical antidepressants encourage synaptic action in the monoamines including dopamine (DA) through blocking the neuronal reuptake of these monoamines [2]. Serra et al. [3] suggested that the involvement of dopaminergic presynaptic receptors in the antidepressants’ activity a long back, it was not mentioned as one of the accepted hypotheses of mental depression.
The impaired antioxidant system is associated with Reactive Oxygen Species (ROS) [4]. An increase in the production of ROS in depression is associated with increased levels of malondialdehyde and arachidonic acid [5]. It was reported an increase in oxidative stress in the animals submitted to stress [5]. It is accepted to use antioxidant agents for treatment of the depression [6].
Menthol is a strong antioxidant and improves the antioxidant and lipid profile in animals [7]. Abbasi-Maleki et al. [8] have reported that crude extract of Mentha piperita showed antidepressant-like effect through the monoaminergic system due to its compound such as menthol. As far as we know, no scientific study is conducted to evaluate the antidepressant-like effects of menthol by forced swimming test (FST) and through dopaminergic system using a mice model. The FST is broadly used in pharmacological models for evaluating the antidepressant effects [8].
The study was thus performed for evaluating the antidepressant-like effects of menthol via dopaminergic system in the FST model.
Materials and Methods
Animals
Male Swiss albino mice with weight of 22±2g and 6 weeks of age were used in this study. The animals were maintained in cages (n=4) with a lighting cycle 12h light/12h dark cycle, ambient temperature of 23-25°C and humidity of 55±15%. The feed and water were given ad libitum. The commercial pellet food was bought from Javaneh Khorasan, Iran.
Materials
Menthol, SCH 23390 and sulpiride were purchased from Sigma-Aldrich, St. Louis, MO, USA. Only SCH 23390 was subcutaneously (SC) administrated, but other agents and menthol were intra-peritoneally (IP) administered in a constant volume of 10ml/kg. The FST was conducted 1h after the single injection of the chemicals and menthol.
Acute toxicity test
We used the Lorke's method for investigating toxicity test [9]. Briefly, after 24h of fasting, a total number of twenty NMRI mice were divided into 5 groups (n=4) and intra-peritoneally treated with vehicle (normal saline with 10% Tween-80; 10ml/kg) and menthol (10, 15, 20, and 30mg/kg). The animals were monitored for behavioral and toxicity signs and also mortality for 24h.
Forced Swimming Test
The FST as a pharmacological test is used for evaluating the antidepressant activity of the agents. We conducted the FST as reported by Porsolt et al. [10]. 28 NMRI mice were grouped into seven groups as follows:
- Group I = vehicle (with 10% Tween 80; 10ml/kg, IP);
- Groups II-V = menthol (10, 15, 20, and 30mg/kg; IP, respectively); and
- Groups VI-VII = fluoxetine (20mg/kg, IP) and imipramine (30mg/kg, IP)
The immobility, swimming and climbing were evaluated as stated by previous studies [8] for the rest four minutes during a six-minute period. We considered decreased immobility time and increased climbing or swimming times as behavioral profiles consistent with antidepressant-like activity [10].
Dopaminergic system of menthol antidepressant-like
To evaluate the potential involvement of the dopaminergic system in the antidepressant-like activity of menthol, the mice, were pre-treated with SCH 23390 (0.05mg/kg, SC, a dopamine D1 receptor antagonist), sulpiride (50mg/kg, IP, a dopamine D2 receptor antagonist) and haloperidol (0.2mg/kg, IP, non-selective dopamine receptor antagonist) 15min before the IP administration of menthol (20mg/kg, IP) or vehicle (10ml/kg, IP). The animals were exposed to FST 1h after the treatment [8].
Statistical analysis
The data were analyzed by the one-way ANOVA for dose-response curves and time-course curves and two-way for assessing of the mechanism of action followed by Tukey's post-hoc test when appropriate.
Findings
Acute toxicity test
There were no any mortality and toxicity signs in the mice studied with doses used (10-30mg/kg, IP).
Immobility behavior
Our findings for one-way ANOVA in Figure1 showed a significant reduction in immobility time (p<0.001) and a significant increase in swimming time (p<0.001) in the mice treated with menthol compared with the vehicle group. There were significant differences in the immobility time caused by menthol compared to that produced by treatment of imipramine and fluoxetine. The one-way ANOVA did not also show significant differences between the mice treated with menthol (20 and 30mg/kg, IP) and fluoxetine in swimming time (p>0.05) and also between the animals administrated with menthol (30mg/kg, IP) and imipramine in climbing time (p>0.05). The best response was observed in dose of the 20 and 30mg/kg dose, this dose was thus used 20mg/kg dose for the subsequent tests (Figure 1).
Involvement of the dopaminergic system in the antidepressant-like effect of mentholin the FST
Pre-treatment of the animals with SCH 23390 (a dopamine D1 receptor antagonist), sulpiride (a dopamine D2 receptor antagonist) and haloperidol (non-selective dopamine receptor antagonist) significantly prevented the antidepressant-like effect of menthol (Figure 2).

Figure 1. The effect of the IP administration of the menthol in the FST (****p<0.0001; ***p<0.001)

Figure 2. The effect of the pretreatment of animals with SCH23390, sulpiride and haloperidol on the antidepressant-like effect created by the menthol in the FST (***significant differences among other groups and the control at p<0.001; +++significant differences among other groups and the extract group at p<0.001)
The OFT
It was not observed significant changes (p>0.05) in the crossing (Figure 3) and rearing counts in the animals treated with menthol (10-30mg/kg; IP) in comparison to the vehicle group.

Figure 3. The effect of the menthol in OFT in the mice. Values are expressed as mean ± S.D.
Discussion
This study was conducted to evaluate the antidepressant-like effects of menthol by FST through dopaminergic system in mice model. In the current study, the results showed that menthol administered through i.p. route yields the antidepressant-like effect in the FST in mice. In addition, the locomotor activity was not altered. In addition, an involvement of dopaminergic (D1 and D2 receptors) systems in the antidepressant-like effect of menthol was observed. The FST is commonly used as a behavioral test and also a model for evaluation of the depression in rodents [10]. The results showed that the effect of the menthol at dose of 30mg/kg in the FST was similar to the response of fluoxetine and imipramine. It was reported that typical antidepressants encourage synaptic action in the monoamines including dopamine (DA) through blocking the neuronal reuptake of these monoamines [2]. It has been accepted the essential role of monoaminergic system in the patho- physiology and treatment of depression [2].
Traditional antidepressants decrease the depression signs by increasing the bioavailability of monoamines in the brain [11, 12]. The antidepressants have been used for the treatment of depression, because of increased synaptic levels of these monoamines [13]. It seems that menthol increases the levels of monoamines as is evidenced by increased dopaminergic levels. It has been reported that the antidepressant-like effects of active compounds such as menthone [14], cineole [15] and pulegone [16]. There is no any report available in the literature for antidepressant-like effects of menthol. Abbasi-Maleki et al. [8] attributed antidepressant-like effects of medicinal plants to their antioxidant properties. The results showed that menthol acts in the antidepressant-like effects through dopaminergic system. Antidepressant-like effects of the menthol were reversed through the pretreatment of animals with SCH23390, sulpiride, and haloperidol suggesting that menthol exerts antidepressant-like effects via the dopaminergic system. It has been accepted the role of the D1 and D2 receptors in the treatment of depression, because antagonists of these receptors reverse the antidepressant-like action of some antidepressants [17-19]. The dopamine directly promotes the factors diminished in people with depression such as motivation and attention [20]. It was reported that some dopamine receptor agonists have positive roles in the treatment of refractory and bipolar depression [21]. The results showed that dopamine D1 and D2 receptor antagonist reversed the antidepressant-like effect of the menthol showing that dopamine D1 and D2 receptors mediate the antidepressant-like effect of menthol. The results also showed that pretreatment of mice with haloperidol could reverse the antidepressant-like effect of the menthol, suggesting that dopamine receptors modulate in antidepressant-like response of the menthol.
Conclusion
The administration of menthol exerts antidepressant-like effects through dopaminergic (D1 and D2) system.
Acknowledgements: None declared by the authors.
Ethical Permission: Compliance with ethical guidelines: Approval for this study was obtained from International Center for Intelligent Research.
Conflicts of Interests: None declared by the authors.
Authors' contributions: All authors contributed toward data analysis, drafting and revising the paper and agreed to be responsible for all the aspects of this work.
Funding/Support: This study was supported by a grant from Georgian Center for Neuroscience Research (GCNR-2018-1841335).
References
1. Sedighi S, Nasiri B, Alipoor R, Moradi-Kor N. Modulation of 6-Gingerol in antidepressant-like effects: An investigation of serotonergic system in mice model. GMJ Med. 2017;1(1):21-5. [Persian] [Link] [DOI:10.29088/GMJM.2017.21]
2. Lambert G, Johansson M, Agren H, Friberg P. Reduced brain norepinephrine and dopamine release in treatment-refractory depressive illness: Evidence in support of the catecholamine hypothesis of mood disorders. Arch Gen Psychiatr. 2000;57(8):787-93. [Link] [DOI:10.1001/archpsyc.57.8.787]
3. Serra G, Agriolas A, Klimek V, Fadda F, Gessa GL. Chronic treatment with antidepressants prevents the inhibitory effect of small doses of apomorphine on dopamine synthesis and motor activity. Life Sci. 1979;25(5):415-23. [Link] [DOI:10.1016/0024-3205(79)90573-3]
4. Maes M, Galecki P, Chang YS, Berk M. A review on the oxidative and nitrosative stress (O&NS) pathways in major depression and their possible contribution to the (Neuro) degenerative processes in that illness. Progr Neuro-Psychopharmacol Biol Psychiatry. 2011;35(3):676-92. [Link] [DOI:10.1016/j.pnpbp.2010.05.004]
5. Lucca G, Comim CM, Valvassori SS. Increased oxidative stress in sub mitochondrial particles into the brain of rats submitted to the chronic mild stress paradigm. J Psychiatric Res. 2009;43(9):864-9. [Link] [DOI:10.1016/j.jpsychires.2008.11.002]
6. Scapagnini G, Davinelli S, Drago F, de Lorenzo A, Oriani G. Antioxidants as antidepressants: Fact or fiction. CNS Drug. 2012;26(6):477-90. [Link] [DOI:10.2165/11633190-000000000-00000]
7. Mesbahzadeh B, Garmsiri M, Jalalvand F, Shojaie L, Kakar MA. Oral administration of menthol could alleviate adverse effects of polycystic ovarian syndrome in blood biochemical parameters and antioxidant status in experimental Wister rats. GMJ Med. 2017;1(1):9-14. [Persian] [Link] [DOI:10.29088/GMJM.2017.09]
8. Abbasi-Maleki S, Bakhtiarian A, Nikoui V. Involvement of the monoaminergic system in the antidepressant-like effect of the crude extract of Mentha piperita (Lamiaceae) in the forced swimming test in mice. Synergy. 2017;5:21-8. [Persian] [Link] [DOI:10.1016/j.synres.2017.08.002]
9. Lorke D. A new approach to practical acute toxicity testing. Arch Toxicol. 1983;54(4):275‑87. [Link] [DOI:10.1007/BF01234480]
10. Porsolt RD, Bertin A, Jalfre M. Behavioral despair in mice: A primary screening test antidepressants. Arch Int Pharmacodyn Ther. 1977;229(2):327-36. [Link]
11. Brunello N, Mendlewicz J, Kasper S, Leonard B, Montgomery S, Nelson JC. The role of noradrenaline and selective noradrenaline reuptake inhibition in depression. Eur Neuropsychopharmacol. 2002;12(5):461-75. [Link] [DOI:10.1016/S0924-977X(02)00057-3]
12. Blier P. Possible neurobiological mechanisms underlying faster onset of antidepressant action. J Clin Psychiatry. 2001;62(4):7-11. [Link]
13. Risch SC, Nemeroff CB. Neurochemical alterations of serotonergic neuronal systems in depression. J Clin Psychiatry. 1992;10:3-7. [Link]
14. Xue JS, Li H, Deng X, Fu Q, Ma S. L-Menthone confers antidepressant-like effects in an unpredictable chronic mild stress mouse model via NLRP3 inflammasome-mediated inflammatory cytokines and central neurotransmitters. Pharmacol Biochem Behav. 2015;134:42-8. [Link] [DOI:10.1016/j.pbb.2015.04.014]
15. Gomes B, Feitosa ML, Silva MIG, Noronha EC, Moura BA, Venancio ET, et al. Anxiolytic-like effect of the monoterpene 1, 4-cineole in mice. Pharmacol Biochem Behav. 2010;96(3):287-93. [Link] [DOI:10.1016/j.pbb.2010.05.019]
16. Ozturk Y, Aydm S, Tecik B, Baser K. Effects of essential oils from certain Ziziphora species on swimming performance in mice. Phytother Res. 1995;9:225-7. [Link] [DOI:10.1002/ptr.2650090315]
17. Hirano S, Miyata S, Onodera K, Kamei J. Involvement of dopamine D 1 receptors and a 1-adrenoceptors in the antidepressant-like effect of chlorpheniramine in the mouse tail suspension test. Eur J Pharmacol. 2007;562(1-2):72-6. [Link] [DOI:10.1016/j.ejphar.2007.01.063]
18. Machado DG, Kaster MP, Binfare RW, Dias M, Santos AR, Pizzolatti MG. Antidepressant-like effect of the extract from leaves of Schinus molle L. in mice: evidence for the involvement of the monoaminergic system. Prog Neuropsychopharmacol Biol Psychiatry. 2007;31(2):421-8. [Link] [DOI:10.1016/j.pnpbp.2006.11.004]
19. Yamada J, Sugimoto Y, Yamada S. Involvement of dopamine receptors in the anti-immobility effects of dopamine re-uptake inhibitors in the forced swimming test. Eur J Pharmacol. 2004;504(3):207-11. [Link] [DOI:10.1016/j.ejphar.2004.09.057]
20. Abbasi-Maleki S, Mousavi SZ. Hydroethanolic extract of Carthamus tinctorius induces antidepressant-like effects: Modulation by dopaminergic and serotonergic systems in tail suspension test in mice. Iran J Basic Med Sci. 2017;20(9):1063-73. [Link]
21. Takamura N, Nakagawa S, Masuda T, Boku S, Kato A, Song N. The effect of dopamine on adult hippocampal neurogenesis. Prog Neuropsychopharmacol Biol Psychiatry. 2014;50:116-24. [Link] [DOI:10.1016/j.pnpbp.2013.12.011]









