GMJ Medicine
eISSN : 2626-3041
Volume 3, Issue 2 (2024)
GMJM 2024, 3(2): 57-61 |
Back to browse issues page
Article Type:
Subject:
History
Received: 2023/09/20 | Accepted: 2024/02/18 | Published: 2024/04/7
Received: 2023/09/20 | Accepted: 2024/02/18 | Published: 2024/04/7
How to cite this article
Forghani N, Rahimian Boogar M, Faramarzi H. Mental and Muscle Relaxation in Water Decreases Liver Enzymes and Inflammatory Markers in Non-Alcoholic Fatty Liver Disease. GMJM 2024; 3 (2) :57-61
URL: http://gmedicine.de/article-2-254-en.html
URL: http://gmedicine.de/article-2-254-en.html
Download citation:
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:



Rights and permissions
BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
1- Department of Cell and Molecular Biology, School of Biology, College of Sciences, Tehran University, Tehran, Iran
2- School of Medical, Shiraz University of Medical Sciences, Shiraz, Iran
2- School of Medical, Shiraz University of Medical Sciences, Shiraz, Iran
Keywords:
| Abstract (HTML) (2355 Views)
Full-Text: (808 Views)
Introduction
Non-alcoholic fatty liver disease (NAFLD) has increased over the past years. NAFLD occurs when liver cells begin to collect fat droplets (mainly triglycerides). Accumulation of fat in the liver promotes the development of inflammation and progression of fibrosis, which can ultimately lead to patient death. Pathologically, NAFLD is associated with obesity and overweight, type 2 diabetes, hypertension, insulin resistance, and metabolic syndrome [1].
Studies have shown that liver enzymes are the best indicator for assessing liver status [2]. Elevations of alanine aminotransferase (ALT) and aspartate aminotransferase (AST) may be due to NAFLD. Serum levels of ALT enzyme are higher than AST in NAFLD patients [3]. Among the enzymes in the liver, the ALT enzyme has been associated with inflammation from fat accumulation in the liver more than others [4]. NAFLD is influenced by age, gender, ethnicity, and endocrine dysfunctions (hypothyroidism, hypopituitarism, hypogonadism, and polycystic ovary syndrome) [5, 6]. NAFLD has a direct relationship with obesity. 80% of obese people suffer from NAFLD [7, 8]. The incidence of this disease in women is higher than in men, although opposite results have been reported [9]. NAFLD patients have an unhealthy lifestyle in both diet and physical activity [10]. Regarding the pathology of non-alcoholic liver disease, treatment has not yet been completely and specifically developed for this disease [11]. Many previous studies have shown on human specimens that regular exercise and diet play an important role in improving insulin resistance and other important factors involved in the NAFLD pathogens, such as hypertriglyceridemia, hyperglycemia, syndrome metabolism, obesity, and the reduction of AST and ALT enzymes [12]. Studies have also been done on the role of vitamin D in improving the symptoms of fatty liver disease. In their opinion, Vitamin D supplements might improve NAFLD by inflammation reduction [13]. The accumulation of fat -both in the adipose tissue and in liver steatosis- leads to increased CRP (C-reactive protein) levels among obese patients. Thus, CRP may be a marker of NAFLD in obese patients. CRP was reported to increase in metabolic syndrome and type-2 diabetes [14]. C-reactive protein (CRP) is made in the liver. This protein is known as acute phase protein. Its level rises when there is inflammation in the body. Some studies have shown that CRP increases in NAFLD [15]. Cytokines play a pivotal active role in NAFLD and are considered potential therapeutic targets. Among cytokines, Interleukin 6 promotes the synthesis of acute-phase proteins such as CRP. It can be said that IL-6 plays a role in the pathogenesis of NAFLD. Serum levels of interleukin6 are high in people with high fatty liver [16]. IL-6 mediates the synthesis of acute-phase proteins such as C-reactive protein [17]. Serum levels of IL-6 are higher in patients with NAFLD [18, 19]. Due to these characteristics, the role of IL-6 in the pathogenesis of fatty liver patients cannot be ignored. Perhaps this role is indirect. Using water as a therapy is one of the oldest treatments in natural medicine, which is called water therapy or hydrotherapy [20]. Studies have shown the effect of hydrotherapy on various systems of the body, such as the management of pain, chronic obstructive pulmonary diseases, anorectal disorders, fatigue, anxiety, obesity, hypercholesterolemia, and hyperthermia.
In the present study, the effect of mental and muscle relaxation in water was investigated with the aim of evaluating changes in liver enzymes and inflammatory markers (such as IL-6 and CRP) in NAFLD patients.
Materials and Methods
Study design
The present study was a randomized clinical trial with pretest-posttest and follow-up with a control group. The independent variable was water therapy. The dependent variables were ALT, AST, and ALP liver enzymes and inflammatory markers (such as IL-6 and CRP). Clinical trials were conducted at Dr. Zahedi's lab in Larestan City in 2018(May to November). Ethical consent was obtained from individuals who participated in this research. Inclusion criteria were NAFLD disease with grade I, obesity and overweight, gender female, age 15 to 65 years, Body mass index (BMI) 25 to 30 or higher, and mental and physical health for learning and exercising. Patients with any of the following conditions were excluded: Smoking and drinking, Opioid drug addiction, Consumption of industrial psychosocial drugs, Cardiovascular disease, viral hepatitis B and C disease, Respiratory diseases that could lead to shortness of breath in the water, Sensitivity, and skin ulcers, Inability to learn swimming skills (at the elementary level), Fear of water, Physical disability and the presence of symptoms of mental disorders.
Method of treatment protocols
In order to determine the level of liver enzymes (ALT, AST, and ALP), as well as IL-6 and CRP, 10cc of the fasting blood sample were taken from the anterior venous of the patients. There were two groups. Non-NAFLD patients (n=10) and NAFLD patients (n=10). The enzyme kinetic method and ALT, AST, and ALP enzyme levels were measured to determine liver enzymes. First, mixed substrates and coenzymes were placed in bain marie for 10 to 15 minutes; then, by adding a serum, enzymes were measured and recorded in 4 minutes using a photometer. Serum levels of IL-6 and CRP were measured with an ELISA kit.
Mental and Muscle relaxation test in water
In this study, 4 types of walking in the water were performed every other day, and each session was performed for 60 minutes in 8 weeks, with the training intensity being 55-60% of the VO2max. Four types of hydrotherapy exercises included moving the marsh, walking on the heel of the foot, walking on the toe, and butterfly swimming. Immediately after the completion of therapeutic courses, 10cc of fasting blood samples were taken from the patients, and the parameters were measured and recorded as post-test. The post-test stage also measured and recorded weight and body mass index.
Data analysis
The data were analyzed using SPSS 25. Means were compared with paired t-test and two-way mixed ANOVA (repeated measures) for FBS, ALT, AST, ALP, CRP, and IL-6 before and after each group's mental and muscle relaxation in water (within-group and between-group). The independent samples t‑test was used to evaluate the difference between the baseline characteristics of study participants in two groups. Also, the correlation test was used to evaluate the correlation between age and FBS with liver enzymes.
Findings
Twenty of 40 volunteers completed the study, and twenty participants discontinued it for some reason. Seventeen participants (eight in the treatment group and nine in the control group) withdrew during the study period for personal reasons, and three participants (two in the treatment group and one in the control group) did not follow up. Thus, the main analyses were conducted with 20 participants (treatment group, n=10; control group, n=10).
Baseline characteristics of the patients
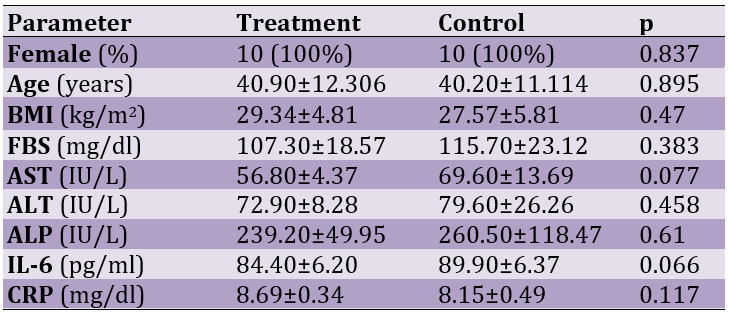
The median age of the patients was 40.9±12.3 years. The mean fasting blood glucose (FBS), liver enzymes (ALT and AST), IL-6, and CRP were higher than the standard laboratory scale. The characteristics of the patients showed no significant differences between groups (Table 1).
Table 1. Baseline variables in participants of study groups
The level of liver enzymes increased with increasing age
There was a positive and significant correlation between the AST and ALP liver enzymes with age. So that this relationship was confirmed at a significant level of 95% (p≤0.05). Also, there was a statistically significant correlation between IL-6 and CRP with subjects' age.
FBS can be involved in liver enzyme disorder in NAFLD
Our Results showed that fasting blood glucose (FBS) can predict 38.2% of ALT variance, 38.5% of AST liver enzyme variability, and 35.9% of ALP variability in liver enzymes. Furthermore, the results of this study indicated that whenever the FBS increased, the levels of ALT, AST, and ALP enzymes increased in blood plasma. This means that the FBS variable can be involved in the liver enzyme disorder of NAFLD disease. Analysis results showed there was a significant positive correlation between age and FBS with ALT and AST. There was no significant correlation between FBS and ALP.
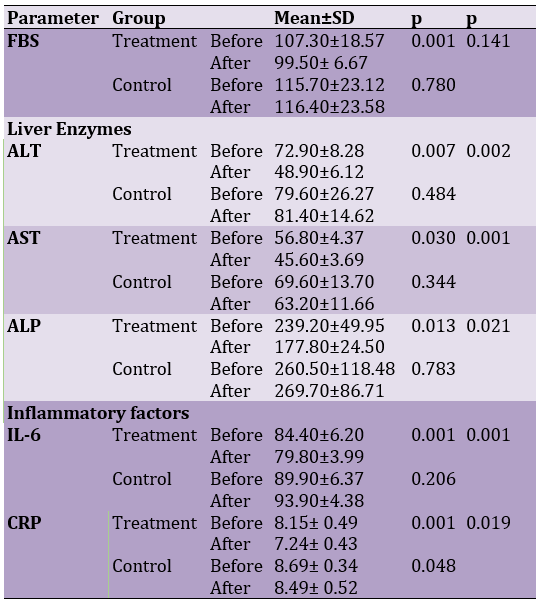
The mean and standard deviation of FBS, liver enzyme levels, and inflammatory markers before and after mental and muscle relaxation were reported in Table 2.
Table 2. Mean and standard deviation of FBS, liver enzymes, and Inflammatory factors in study groups
Discussion
The purpose of this study was to determine the effect of mental and muscle relaxation in water on liver enzymes and inflammatory markers (such as CRP and IL-6), whose value is high in obese, inactive women with non-alcoholic fatty liver disease. In this study, the required data were collected using clinical trials. and parameters included: levels of liver enzymes (ALT, AST, and ALP) also, CRP and IL-6 were measured and recorded in two stages of pre-test and post-test and control group. Finally, the collected data were analyzed by descriptive statistical tests. The findings of this study indicated that the mean FBS, ALT, AST, CRP, and IL-6 in the research group were higher than standard and optimal levels, as has been reported by other studies [21-23]. Also, studies have confirmed the relationship between non-alcoholic fatty liver and fasting blood glucose (FBS). People with high blood sugar usually have a fatty liver [1]. Our results also confirmed this. Liver enzymes show different aspects of the normal functioning of the liver; for example, the normal levels of the ALT or AST enzymes represent the integrity of the liver cells, and the normal levels of the ALP enzyme indicate the production and secretion of sufficient albumin to make the protein [24]. The results of this study indicated that ALT and AST increased with increasing age. Some studies have shown that age significantly affects liver enzymes [25]. Also, in other research, the results of muscle therapy showed that muscle therapy methods have an effective role in reducing stress and headaches in women with migraine headaches [26].
Our results of paired sample t-test and two-way mixed ANOVA (repeated measures) showed that there was a significant difference in reduced levels of ALT, AST, ALP, CRP, and IL-6 after 8 weeks of treatment, which is confirmed at 95% level. Exercise reduces plasma-free fatty acids, apoptosis, and advanced fibrosis [10]. Script Nick et al., in a study aimed at examining the effect of endurance and strength training on biochemical parameters of liver function in women with abdominal obesity, showed that exercising could decrease the level of serum ALP in patients with NAFLD [27]. Also, other research results in 2004 showed that ALT and AST levels decreased after the end of the six-week exercise program [28].
The effect of exercise training on inflammatory factors has been studied. Studies have shown that muscle movement has anti-inflammatory properties [29]. The effect of aerobic and resisted
exercise training on the reduction of inflammatory factors in NAFLD was investigated in research. They observed that significantly decreased the amount of inflammatory factors [30].
Recent data showed that regular exercise effectively reduced liver fat content and improved the diagnostic profile of patients with non-alcoholic fatty liver [31].
Therefore, due to the fact that increased fat stores play an important role in the development of NAFLD and its underlying metabolic diseases, including increased cholesterol and triglycerides, aerobic exercise activities Such as water therapy can have a significant effect on weight loss, BMI, body fat loss, de-aggregation and decrease in triglyceride accumulation in the liver, decrease in sedimentation and total cholesterol and LDL accumulation in the liver.
According to the studies, exercise reduces fat stores, diseases like cholesterol and triglycerides, diabetes, and metabolic syndrome, thereby reducing the risk of developing NAFLD. On the other hand, to this day, weight loss is the only definitive treatment for non-alcoholic liver disease, and lifestyle interventions are only part of its management. Therefore, exercise activities can potentially be effective in reducing fatty liver.
Conclusion
Mental and muscle relaxation in water (aqua yoga) is one of the gentlest yet most effective forms of water sports. It takes advantage of buoyancy, water suspension, and in-depth stretching and relaxation. It increases respiratory capacity without strain, so patients can relax and stretch at the same time. The novelty of our work relates to the use of water to help patients achieve muscle and mental relaxation in water, which has changed the parameters of our study.
This research indicates that mental and muscle relaxation in water and sports activity is due to increased daily energy intake, improved muscle oxidation, decreased free fatty acid transfusion into the liver, and reduced fatty sedimentation in the liver. As a result, muscle relaxation in water (aqua yoga) plays an important role in improving NAFLD.
Acknowledgments: None declared by the authors.
Ethical Permission: None declared by the authors.
Conflicts of Interests: None declared by the authors.
Funding/Support: None declared by the authors.
Non-alcoholic fatty liver disease (NAFLD) has increased over the past years. NAFLD occurs when liver cells begin to collect fat droplets (mainly triglycerides). Accumulation of fat in the liver promotes the development of inflammation and progression of fibrosis, which can ultimately lead to patient death. Pathologically, NAFLD is associated with obesity and overweight, type 2 diabetes, hypertension, insulin resistance, and metabolic syndrome [1].
Studies have shown that liver enzymes are the best indicator for assessing liver status [2]. Elevations of alanine aminotransferase (ALT) and aspartate aminotransferase (AST) may be due to NAFLD. Serum levels of ALT enzyme are higher than AST in NAFLD patients [3]. Among the enzymes in the liver, the ALT enzyme has been associated with inflammation from fat accumulation in the liver more than others [4]. NAFLD is influenced by age, gender, ethnicity, and endocrine dysfunctions (hypothyroidism, hypopituitarism, hypogonadism, and polycystic ovary syndrome) [5, 6]. NAFLD has a direct relationship with obesity. 80% of obese people suffer from NAFLD [7, 8]. The incidence of this disease in women is higher than in men, although opposite results have been reported [9]. NAFLD patients have an unhealthy lifestyle in both diet and physical activity [10]. Regarding the pathology of non-alcoholic liver disease, treatment has not yet been completely and specifically developed for this disease [11]. Many previous studies have shown on human specimens that regular exercise and diet play an important role in improving insulin resistance and other important factors involved in the NAFLD pathogens, such as hypertriglyceridemia, hyperglycemia, syndrome metabolism, obesity, and the reduction of AST and ALT enzymes [12]. Studies have also been done on the role of vitamin D in improving the symptoms of fatty liver disease. In their opinion, Vitamin D supplements might improve NAFLD by inflammation reduction [13]. The accumulation of fat -both in the adipose tissue and in liver steatosis- leads to increased CRP (C-reactive protein) levels among obese patients. Thus, CRP may be a marker of NAFLD in obese patients. CRP was reported to increase in metabolic syndrome and type-2 diabetes [14]. C-reactive protein (CRP) is made in the liver. This protein is known as acute phase protein. Its level rises when there is inflammation in the body. Some studies have shown that CRP increases in NAFLD [15]. Cytokines play a pivotal active role in NAFLD and are considered potential therapeutic targets. Among cytokines, Interleukin 6 promotes the synthesis of acute-phase proteins such as CRP. It can be said that IL-6 plays a role in the pathogenesis of NAFLD. Serum levels of interleukin6 are high in people with high fatty liver [16]. IL-6 mediates the synthesis of acute-phase proteins such as C-reactive protein [17]. Serum levels of IL-6 are higher in patients with NAFLD [18, 19]. Due to these characteristics, the role of IL-6 in the pathogenesis of fatty liver patients cannot be ignored. Perhaps this role is indirect. Using water as a therapy is one of the oldest treatments in natural medicine, which is called water therapy or hydrotherapy [20]. Studies have shown the effect of hydrotherapy on various systems of the body, such as the management of pain, chronic obstructive pulmonary diseases, anorectal disorders, fatigue, anxiety, obesity, hypercholesterolemia, and hyperthermia.
In the present study, the effect of mental and muscle relaxation in water was investigated with the aim of evaluating changes in liver enzymes and inflammatory markers (such as IL-6 and CRP) in NAFLD patients.
Materials and Methods
Study design
The present study was a randomized clinical trial with pretest-posttest and follow-up with a control group. The independent variable was water therapy. The dependent variables were ALT, AST, and ALP liver enzymes and inflammatory markers (such as IL-6 and CRP). Clinical trials were conducted at Dr. Zahedi's lab in Larestan City in 2018(May to November). Ethical consent was obtained from individuals who participated in this research. Inclusion criteria were NAFLD disease with grade I, obesity and overweight, gender female, age 15 to 65 years, Body mass index (BMI) 25 to 30 or higher, and mental and physical health for learning and exercising. Patients with any of the following conditions were excluded: Smoking and drinking, Opioid drug addiction, Consumption of industrial psychosocial drugs, Cardiovascular disease, viral hepatitis B and C disease, Respiratory diseases that could lead to shortness of breath in the water, Sensitivity, and skin ulcers, Inability to learn swimming skills (at the elementary level), Fear of water, Physical disability and the presence of symptoms of mental disorders.
Method of treatment protocols
In order to determine the level of liver enzymes (ALT, AST, and ALP), as well as IL-6 and CRP, 10cc of the fasting blood sample were taken from the anterior venous of the patients. There were two groups. Non-NAFLD patients (n=10) and NAFLD patients (n=10). The enzyme kinetic method and ALT, AST, and ALP enzyme levels were measured to determine liver enzymes. First, mixed substrates and coenzymes were placed in bain marie for 10 to 15 minutes; then, by adding a serum, enzymes were measured and recorded in 4 minutes using a photometer. Serum levels of IL-6 and CRP were measured with an ELISA kit.
Mental and Muscle relaxation test in water
In this study, 4 types of walking in the water were performed every other day, and each session was performed for 60 minutes in 8 weeks, with the training intensity being 55-60% of the VO2max. Four types of hydrotherapy exercises included moving the marsh, walking on the heel of the foot, walking on the toe, and butterfly swimming. Immediately after the completion of therapeutic courses, 10cc of fasting blood samples were taken from the patients, and the parameters were measured and recorded as post-test. The post-test stage also measured and recorded weight and body mass index.
Data analysis
The data were analyzed using SPSS 25. Means were compared with paired t-test and two-way mixed ANOVA (repeated measures) for FBS, ALT, AST, ALP, CRP, and IL-6 before and after each group's mental and muscle relaxation in water (within-group and between-group). The independent samples t‑test was used to evaluate the difference between the baseline characteristics of study participants in two groups. Also, the correlation test was used to evaluate the correlation between age and FBS with liver enzymes.
Findings
Twenty of 40 volunteers completed the study, and twenty participants discontinued it for some reason. Seventeen participants (eight in the treatment group and nine in the control group) withdrew during the study period for personal reasons, and three participants (two in the treatment group and one in the control group) did not follow up. Thus, the main analyses were conducted with 20 participants (treatment group, n=10; control group, n=10).
Baseline characteristics of the patients
The median age of the patients was 40.9±12.3 years. The mean fasting blood glucose (FBS), liver enzymes (ALT and AST), IL-6, and CRP were higher than the standard laboratory scale. The characteristics of the patients showed no significant differences between groups (Table 1).
Table 1. Baseline variables in participants of study groups

The level of liver enzymes increased with increasing age
There was a positive and significant correlation between the AST and ALP liver enzymes with age. So that this relationship was confirmed at a significant level of 95% (p≤0.05). Also, there was a statistically significant correlation between IL-6 and CRP with subjects' age.
FBS can be involved in liver enzyme disorder in NAFLD
Our Results showed that fasting blood glucose (FBS) can predict 38.2% of ALT variance, 38.5% of AST liver enzyme variability, and 35.9% of ALP variability in liver enzymes. Furthermore, the results of this study indicated that whenever the FBS increased, the levels of ALT, AST, and ALP enzymes increased in blood plasma. This means that the FBS variable can be involved in the liver enzyme disorder of NAFLD disease. Analysis results showed there was a significant positive correlation between age and FBS with ALT and AST. There was no significant correlation between FBS and ALP.
The mean and standard deviation of FBS, liver enzyme levels, and inflammatory markers before and after mental and muscle relaxation were reported in Table 2.
Table 2. Mean and standard deviation of FBS, liver enzymes, and Inflammatory factors in study groups

Discussion
The purpose of this study was to determine the effect of mental and muscle relaxation in water on liver enzymes and inflammatory markers (such as CRP and IL-6), whose value is high in obese, inactive women with non-alcoholic fatty liver disease. In this study, the required data were collected using clinical trials. and parameters included: levels of liver enzymes (ALT, AST, and ALP) also, CRP and IL-6 were measured and recorded in two stages of pre-test and post-test and control group. Finally, the collected data were analyzed by descriptive statistical tests. The findings of this study indicated that the mean FBS, ALT, AST, CRP, and IL-6 in the research group were higher than standard and optimal levels, as has been reported by other studies [21-23]. Also, studies have confirmed the relationship between non-alcoholic fatty liver and fasting blood glucose (FBS). People with high blood sugar usually have a fatty liver [1]. Our results also confirmed this. Liver enzymes show different aspects of the normal functioning of the liver; for example, the normal levels of the ALT or AST enzymes represent the integrity of the liver cells, and the normal levels of the ALP enzyme indicate the production and secretion of sufficient albumin to make the protein [24]. The results of this study indicated that ALT and AST increased with increasing age. Some studies have shown that age significantly affects liver enzymes [25]. Also, in other research, the results of muscle therapy showed that muscle therapy methods have an effective role in reducing stress and headaches in women with migraine headaches [26].
Our results of paired sample t-test and two-way mixed ANOVA (repeated measures) showed that there was a significant difference in reduced levels of ALT, AST, ALP, CRP, and IL-6 after 8 weeks of treatment, which is confirmed at 95% level. Exercise reduces plasma-free fatty acids, apoptosis, and advanced fibrosis [10]. Script Nick et al., in a study aimed at examining the effect of endurance and strength training on biochemical parameters of liver function in women with abdominal obesity, showed that exercising could decrease the level of serum ALP in patients with NAFLD [27]. Also, other research results in 2004 showed that ALT and AST levels decreased after the end of the six-week exercise program [28].
The effect of exercise training on inflammatory factors has been studied. Studies have shown that muscle movement has anti-inflammatory properties [29]. The effect of aerobic and resisted
exercise training on the reduction of inflammatory factors in NAFLD was investigated in research. They observed that significantly decreased the amount of inflammatory factors [30].
Recent data showed that regular exercise effectively reduced liver fat content and improved the diagnostic profile of patients with non-alcoholic fatty liver [31].
Therefore, due to the fact that increased fat stores play an important role in the development of NAFLD and its underlying metabolic diseases, including increased cholesterol and triglycerides, aerobic exercise activities Such as water therapy can have a significant effect on weight loss, BMI, body fat loss, de-aggregation and decrease in triglyceride accumulation in the liver, decrease in sedimentation and total cholesterol and LDL accumulation in the liver.
According to the studies, exercise reduces fat stores, diseases like cholesterol and triglycerides, diabetes, and metabolic syndrome, thereby reducing the risk of developing NAFLD. On the other hand, to this day, weight loss is the only definitive treatment for non-alcoholic liver disease, and lifestyle interventions are only part of its management. Therefore, exercise activities can potentially be effective in reducing fatty liver.
Conclusion
Mental and muscle relaxation in water (aqua yoga) is one of the gentlest yet most effective forms of water sports. It takes advantage of buoyancy, water suspension, and in-depth stretching and relaxation. It increases respiratory capacity without strain, so patients can relax and stretch at the same time. The novelty of our work relates to the use of water to help patients achieve muscle and mental relaxation in water, which has changed the parameters of our study.
This research indicates that mental and muscle relaxation in water and sports activity is due to increased daily energy intake, improved muscle oxidation, decreased free fatty acid transfusion into the liver, and reduced fatty sedimentation in the liver. As a result, muscle relaxation in water (aqua yoga) plays an important role in improving NAFLD.
Acknowledgments: None declared by the authors.
Ethical Permission: None declared by the authors.
Conflicts of Interests: None declared by the authors.
Funding/Support: None declared by the authors.
References
1. Musso G, Gambino R, Cassader M. Recent insights into hepatic lipid metabolism in non-alcoholic fatty liver disease (NAFLD). Prog Lipid Res. 2009;48(1):1-26. [Link] [DOI:10.1016/j.plipres.2008.08.001]
2. FerreiraVS, Pernambuco RB, Lopes EP, Morais C, Rodrigues MC, Arruda M, et al. Frequency and risk factors associated with non-alcoholic fatty liver disease in patients with type 2 diabetesmellitus. Arq Bras Endocrinol Metab. 2010;54(4):362-8. [Link] [DOI:10.1590/S0004-27302010000400004]
3. Neuman MG, Malnick S, Maor Y, Nanau RM, Melzer E, Ferenci P, et al. Alcoholic liver disease: Clinical and translational research. Exp Mol Pathol. 2015;99(3):596-610. [Link] [DOI:10.1016/j.yexmp.2015.09.001]
4. Chen ZW, Chen LY, Dai HL, Chen JH, Fang LZ. Relationship between alanine aminotransferase levels and metabolic syndrome in nonalcoholic fatty liver disease. J Zhejiang Univ Sci B. 2008;9(8):616-22. [Link] [DOI:10.1631/jzus.B0720016]
5. Loria P, Carulli L, Bertolotti M, Lonardo A. Endocrine and liver interaction: The role of endocrine pathways in NASH. Nat Rev Gastroenterol Hepatol. 2009;6(4):236-47. [Link] [DOI:10.1038/nrgastro.2009.33]
6. Vernon G, Baranova A, Younossi ZM. Systematic review: The epidemiology and natural history of non-alcoholic fatty liver disease and non-alcoholic steatohepatitis in adults. Aliment Pharmacol Ther. 2011;34:274-85. [Link] [DOI:10.1111/j.1365-2036.2011.04724.x]
7. Williams CD, Stengel J, Asike MI, Torres DM, Shaw J, Contreras M, et al. Prevalence of nonalcoholic fatty liver disease and nonalcoholic steatohepatitis among a largely middle-aged population utilizing ultrasound and liver biopsy: A prospective study. Gastroenterology. 2011;140:124-31. [Link] [DOI:10.1053/j.gastro.2010.09.038]
8. Bellentani S, Saccoccio G, Masutti F, Crocè LS, Brandi G, Sasso F, et al. Prevalence of and risk factors for hepatic steatosis in Northern Italy. Ann Intern Med. 2000;132(2):112-7. [Link] [DOI:10.7326/0003-4819-132-2-200001180-00004]
9. Lonardo A, Carani C, Carulli N, Loria P. 'Endocrine NAFLD' a hormonocentric perspective of nonalcoholic fatty liver disease pathogenesis. J Hepatol. 2006;44(6):1196-207. [Link] [DOI:10.1016/j.jhep.2006.03.005]
10. Ratziu V, Goodman Z, Sanyal A. Current efforts and trends in the treatment of NASH. J Hepatol. 2015;62:S65-S76. [Link] [DOI:10.1016/j.jhep.2015.02.041]
11. Akyüz F, Demir K, Özdil S, Aksoy N, Poturoğlu Ş, İbrişim D, et al. The effects of rosiglitazone, metformin, and diet with exercise in nonalcoholic fatty liver disease. Dig Dis Sci. 2007;52(9):2359-67. [Link] [DOI:10.1007/s10620-006-9145-x]
12. Okita M, Hayashi M, Sasagawa T, Takagi K, Suzuki K, Kinoyama S, et al. Effect of a moderately energy-restricted diet on obese patients with fatty liver. Nutrition. 2001;17(7-8):542-7. [Link] [DOI:10.1016/S0899-9007(01)00543-3]
13. Hariri M, Zohdi S. Effect of vitamin d on non-alcoholic fatty liver disease: A systematic review of randomized controlled clinical trials. Int J Prev Med. 2019;10:14. [Link] [DOI:10.4103/ijpvm.IJPVM_499_17]
14. Oruc N, Ozutemiz O, Yuce G, Akarca US, Ersoz G, Gunsar F, et al. Serum procalcitonin and CRP levels in non-alcoholic fatty liver disease: a case control study. BMC Gastroenterology. 2009;9:16. [Link] [DOI:10.1186/1471-230X-9-16]
15. Targher G, Bertolini L, Rodella S, Lippi G, Franchini M, Zoppini G, et al. NASH predicts plasma inflammatory biomarkers independently of visceral fat in men. Obesity (Silver Spring). 2008;16(6):1394-9. [Link] [DOI:10.1038/oby.2008.64]
16. Braunersreuther V, Viviani GL, Mach F, Montecucco F. Role of cytokines and chemokines in non-alcoholic fatty liver disease. World J Gastroenterol. 2012;18(8):727-35. [Link] [DOI:10.3748/wjg.v18.i8.727]
17. Yudkin JS, Kumari M, Humphries SE, Mohamed-Ali V. Inflammation, obesity, stress and coronary heart disease: Is interleukin-6 the link? Atherosclerosis. 2000;148(2):209-14. [Link] [DOI:10.1016/S0021-9150(99)00463-3]
18. Kugelmas M, Hill DB, Vivian B, Marsano L, McClain CJ. Cytokines and NASH: A pilot study of the effects of lifestyle modification and vitamin E. Hepatology. 2003;38(2):413-9. [Link] [DOI:10.1053/jhep.2003.50316]
19. Haukeland JW, Damås JK, Konopski Z, Løberg EM, Haa land T, Goverud I, et al. Systemic inflammation in nonalcoholic fatty liver disease is characterized by elevated levels of CCL2. J Hepatol. 2006;44(6):1167-74. [Link] [DOI:10.1016/j.jhep.2006.02.011]
20. Mooventhan A, Nivethitha L. Scientific evidence-based effects of hydrotherapy on various systems of the body. N Am J Med Sci. 2014;6(5):199-209. [Link] [DOI:10.4103/1947-2714.132935]
21. McPherson RA, Pincus MR. Henry's clinical diagnosis and management by laboratory methods. Elsevier Health Sciences; 23rd ed. 2016:P1584. [Link]
22. Androlli TE. Principles of the Internal Medicine of Cecil. Tehran: Taymoor Zadeh Publications, 2010. [Persian] [Link]
23. Ferolla SM, Armiliato G N, Couto CA, Ferrari TC. Probiotics as a complementary therapeutic approach in nonalcoholic fatty liver disease. World J Hepatol. 2015;7(3):559-65. [Link] [DOI:10.4254/wjh.v7.i3.559]
24. Rahmioglu N, Andrew T, Cherkas L, Surdulescu G, Swaminathan R, Spector T, et al. Epidemiology and genetic epidemiology of the liver function test proteins. PLoS One. 2009;4(2):e4435. [Link] [DOI:10.1371/journal.pone.0004435]
25. Amarapurkar D, Kamani P, Patel N, Gupte P, Kumar P, Agal S, et al. Prevalence of non-alcoholic fatty liver disease: Population based study. Ann Hepatol. 2007;6(3):161-3. [Link] [DOI:10.1016/S1665-2681(19)31922-2]
26. Sekhavati E, Rahimian M, Afkari R, Kasharafifard S, Parsaei MV, Lotfi MF, et al. The study of effectiveness of progressive muscle relaxation to reduce symptoms in women with migraine headache. R J Med Sci. 2016;10(3):175-84. [Link]
27. Skrypnik D, Ratajczak M, Karolkiewicz J, Mądry E, Pupek-Musialik D, Hansdorfer-Korzon, et al. Effects of endurance and endurance-strength exercise on biochemical parameters of liver function in women with abdominal obesity. Biomed Pharmacother. 2016;80:1-7. [Link] [DOI:10.1016/j.biopha.2016.02.017]
28. Chitturi S, Farrell GC, George J. Non‐alcoholic steatohepatitis in the Asia-Pacific region: Future shock? J Gastroenterol Hepatol. 2004;19(4):368-74. [Link] [DOI:10.1111/j.1440-1746.2003.03252.x]
29. Petersen AM, Pedersen BK. The anti-inflammatory effect of exercise. J Appl Physiol. 2005;98(4):1154-62. [Link] [DOI:10.1152/japplphysiol.00164.2004]
30. Abd El-Kader SM, Al-Jiffri OH, Al-Shreef FM. Markers of liver function and inflammatory cytokines modulation by aerobic versus resisted exercise training for nonalcoholic steatohepatitis patients. Afr Health Sci. 2014;14(3):551-7. [Link] [DOI:10.4314/ahs.v14i3.8]
31. Sullivan S, Kirk EP, Mittendorfer B, Patterson BW, Klein S. Randomized trial of exercise effect on intrahepatic triglyceride content and lipid kinetics in nonalcoholic fatty liver disease. Hepatol. 2012;55(6):1738-45. [Link] [DOI:10.1002/hep.25548]









